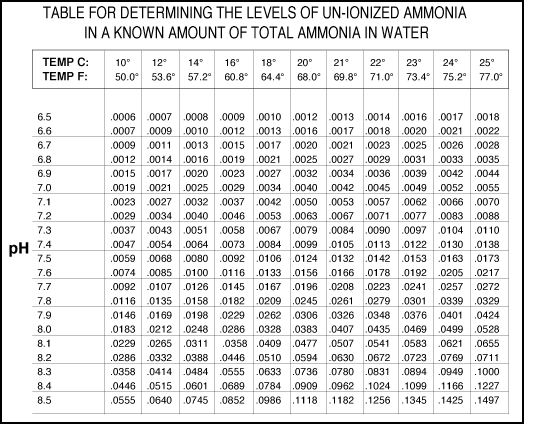
UN-IONIZED AMMONIA (NH3) TABLE
To determine the amount of the
toxic (un-ionized) form of ammonia (NH3)
present in a given amount of total ammonia:
1- Using the appropriate water test kit, determine the amount of
total ammonia ion in your
aquarium or pond (bold face type on the comparator strip).
2- Determine the temperature and pH of the water.
3- Refer to the accompanying table and locate the numerical value
that corresponds to the
temperature and pH of your water.
4- Multiply this number by the reading for total ammonia (from
step 1). The result is the
amount of toxic un-ionized ammonia (in mg/L) in the aquarium or
pond.

Example: the pH is 7.0, the
temperature is 18° C and the
total ammonia reading is .8 mg/L.
Locate the appropriate numerical value (in this example it is .0034).
Multiply .8 times .0034;
the result is .00272. This means that there is about .003 mg/L of
NH3 in this example.
Click here to return to AQUABAZ Home Page